PTEN Target Analysis Report Summary


About the Target
PTEN is a protein that plays a crucial role in various cellular processes. Several studies have investigated the function and regulation of PTEN in different contexts.
One viewpoint is that PTEN can inhibit glucose uptake in cells by blocking the recycling of the glucose transporter GLUT1 to the plasma membrane [1]. This process is mediated by PTEN's interaction with SNX27 and VPS26 retromer complex.
In breast cancer cells, overexpression of a protein called IRIS can repress the transcription of the PTEN gene, resulting in low levels of PTEN protein. This, in turn, activates AKT and inhibits HIF-1alpha phosphorylation, leading to the accumulation of HIF-1alpha and the expression of its target genes involved in metastasis [2].
In another study, it was found that UBE2E1 can ubiquitinate PTEN and mark it for degradation. Knockdown of UBE2E1 rescues PTEN levels, suggesting that UBE2E1 plays a role in PTEN stability [3].
The methylation status of the SALL2 gene can also affect the expression of PTEN in breast cancer cells. Hypomethylation of SALL2 leads to PTEN upregulation, inhibiting Akt/mTOR signaling and resulting in estrogen-dependent growth and tamoxifen sensitivity. Conversely, hypermethylation of SALL2 represses PTEN expression, activating Akt/mTOR signaling and leading to estrogen-independent tumor growth and tamoxifen resistance [4].
Finally, PTEN, along with PLCXD, counteracts endosomal accumulation of a lipid called PtdIns(4,5)P2 in the absence of OCRL. PTEN increases PtdIns(4,5)P2 levels, while OCRL and PLC directly decrease these levels. Thus, the balance between PTEN, OCRL, and PLCXD is crucial for regulating PtdIns(4,5)P2 metabolism and maintaining normal cellular function [5].
Overall, PTEN's diverse functions in cellular processes, including glucose uptake, metastasis, stability regulation, and lipid metabolism, highlight its significance in various biological contexts. [1][2][3][4][5]
From the provided context information, the following key viewpoints can be extracted regarding PTEN:
PTEN loss or inhibition, as well as activation of AKT, can activate PI3K signaling, potentially repressing the expression of MHC molecules and their induction by IFN-gamma. This repression may hinder T-cell activation, tumor recognition, and anti-tumor immunity [6].
Inhibition of PTEN can lead to increased stability of IP3R3, promoting apoptosis in response to IP3 production. PTEN competes with FBXL2 for IP3R3 binding, enhancing its stability and mitochondrial Ca2+ response, ultimately facilitating apoptosis [7].
Ectopic overexpression of FDPS in PTEN-deficient prostate cancer cells increases proliferation and growth advantage [8].
Inhibition of ROR1, combined with erlotinib treatment, can decrease AKT/mTOR signaling pathway activity and enhance cell apoptosis while reducing cell proliferation and survival [9].
Upregulation of miR-21 can lead to the downregulation of various target genes including PTEN. PTEN negatively regulates the PI3K/AKT/mTOR pathway, controlling cell survival, growth, and differentiation [10].
These key viewpoints demonstrate the diverse roles and impacts of PTEN in different cellular processes and cancer-related pathways.
Figure [1]
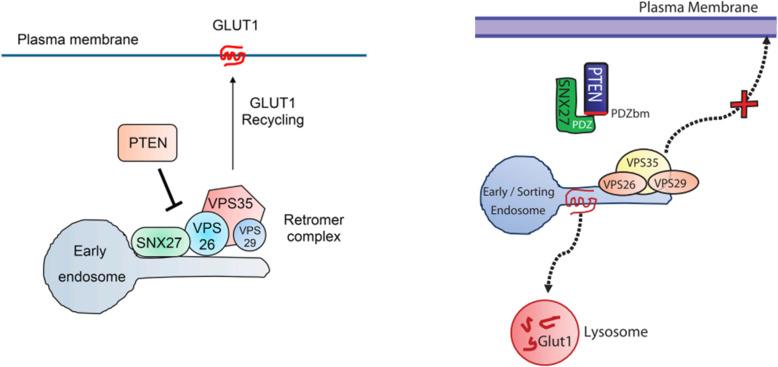
Figure [2]
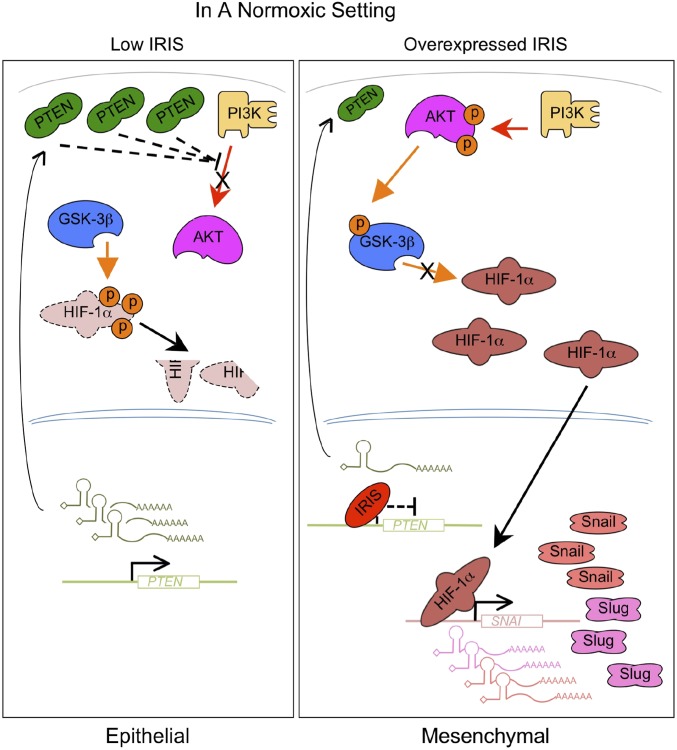
Figure [3]
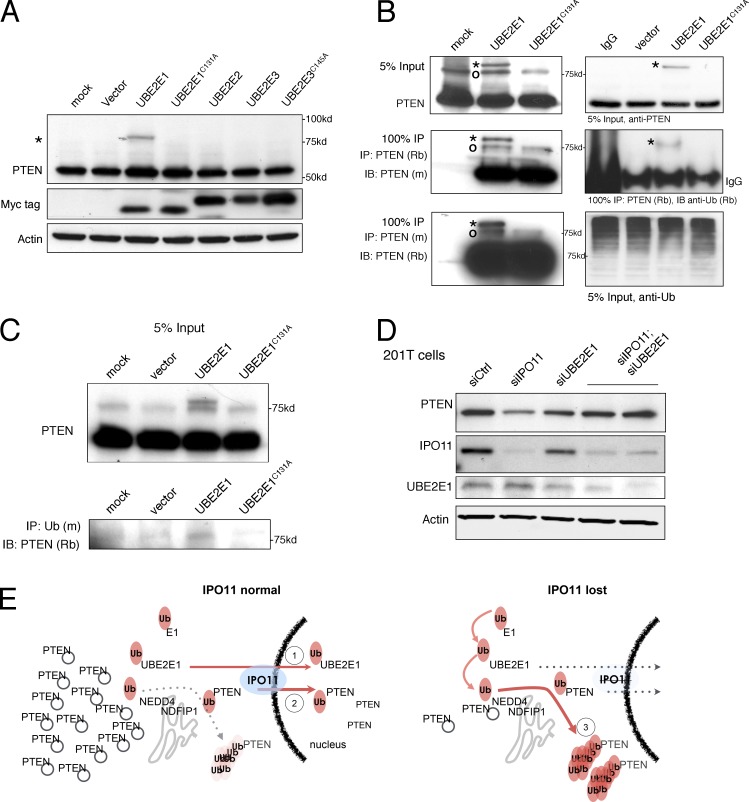
Figure [4]

Figure [5]
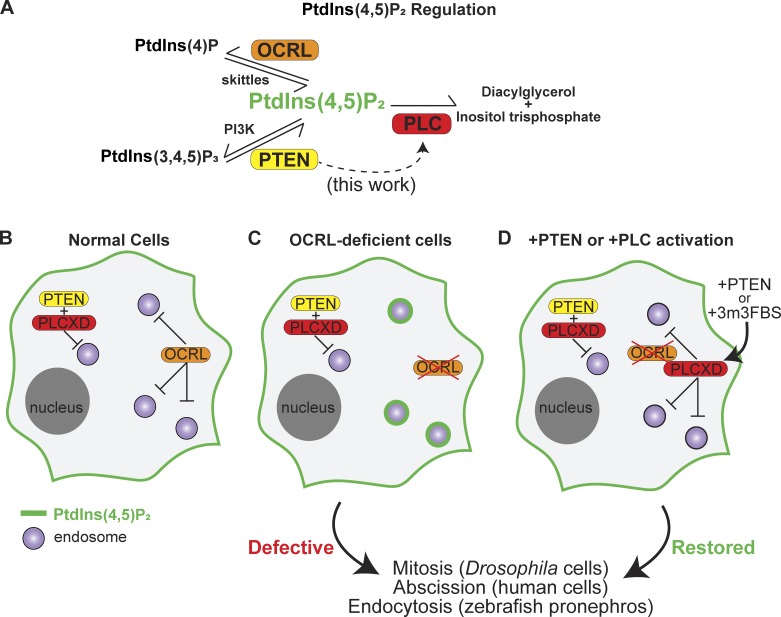
Figure [6]

Figure [7]
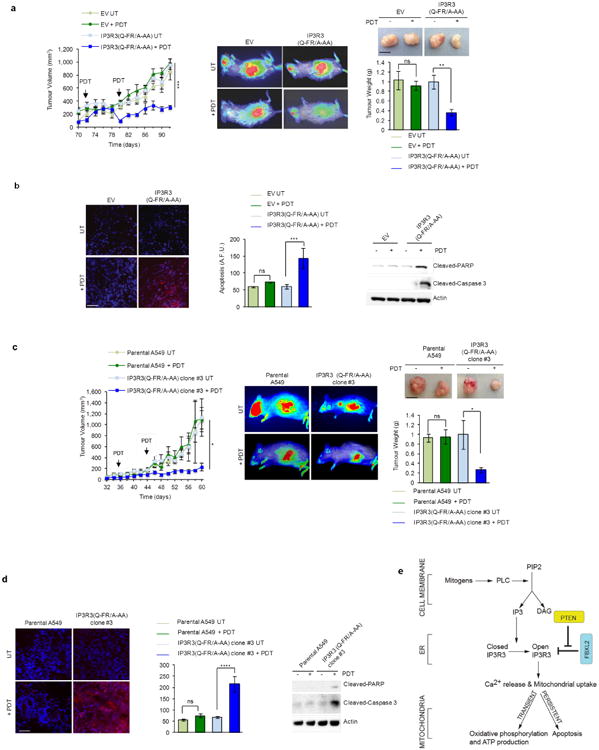
Figure [8]

Figure [9]
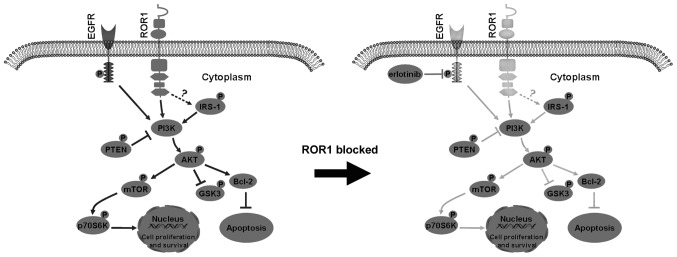
Figure [10]

Note: If you are interested in the full version of this target analysis report, or if you'd like to learn how our AI-powered BDE-Chem can design therapeutic molecules to interact with the PTEN target at a cost 90% lower than traditional approaches, please feel free to contact us at BD@silexon.ai.
More Common Targets
ABCB1 | ABCG2 | ACE2 | AHR | AKT1 | ALK | AR | ATM | BAX | BCL2 | BCL2L1 | BECN1 | BRAF | BRCA1 | CAMP | CASP3 | CASP9 | CCL5 | CCND1 | CD274 | CD4 | CD8A | CDH1 | CDKN1A | CDKN2A | CREB1 | CXCL8 | CXCR4 | DNMT1 | EGF | EGFR | EP300 | ERBB2 | EREG | ESR1 | EZH2 | FN1 | FOXO3 | HDAC9 | HGF | HMGB1 | HSP90AA1 | HSPA4 | HSPA5 | IDO1 | IFNA1 | IGF1 | IGF1R | IL17A | IL6 | INS | JUN | KRAS | MAPK1 | MAPK14 | MAPK3 | MAPK8 | MAPT | MCL1 | MDM2 | MET | MMP9 | MTOR | MYC | NFE2L2 | NLRP3 | NOTCH1 | PARP1 | PCNA | PDCD1 | PLK1 | PRKAA1 | PRKAA2 | PTEN | PTGS2 | PTK2 | RELA | SIRT1 | SLTM | SMAD4 | SOD1 | SQSTM1 | SRC | STAT1 | STAT3 | STAT5A | TAK1 | TERT | TLR4 | TNF | TP53 | TXN | VEGFA | YAP1

