NFE2L2 Target Analysis Report Summary


About the Target
Based on the context information provided, some key viewpoints about Nrf2 (synonymous with NFE2L2) can be extracted:
Nrf2 is regulated by multiple epigenetic mechanisms, including DNA methylation, histone acetylation, and histone methylation. Understanding these regulatory mechanisms is crucial for exploring potential modulators of Nrf2 signaling at different levels of gene expression [1].
Genes associated with the NRF2-mediated oxidative stress response pathway show differential regulation in conditions like aldosterone-producing adenoma (APA) and different zones of the adrenal cortex [2].
Nrf2 plays a role in the saccular-to-alveolar transition in the lung and participates in cellular processes such as antioxidant protection, cell cycle regulation, DNA repair, and mitochondria-related metabolism. Nrf2 deficiency can disrupt these processes and activate inflammatory signaling, potentially leading to lung injury and conditions like bronchopulmonary dysplasia (BPD) [3].
The turnover pathway involving the Keap1/Cul3/Rbx1 complex is not the sole mechanism regulating Nrf2 during RV infection. Additional noncanonical regulatory networks are involved in downregulating the Nrf2/HO-1 axis after the initial hours of infection [4].
The activity of Nrf2 is primarily regulated through its association with the E3 ubiquitin ligase KEAP1. Reactive oxygen species and alkylating compounds can disrupt the KEAP1-Nrf2 complex, leading to Nrf2 translocation to the nucleus and the expression of genes involved in oxidative stress resistance. The inhibition of Nrf2 has also been reported [5].
These viewpoints demonstrate that Nrf2 is a critical regulator of oxidative stress response and cellular processes in various tissues, and its dysregulation can contribute to pathological conditions. Researchers are interested in understanding the epigenetic regulation of Nrf2, identifying modulators of Nrf2 signaling, and exploring its association with diseases like BPD and cancer drug resistance.
Based on the provided context information, here is a key viewpoint about Nrf2 (synonymous with NFE2L2):
NRF2 activation: Nrf2 can be activated by inhibiting GSK3beta, which promotes Nrf2 phosphorylation, nuclear exit, and degradation, resulting in reduced antioxidant gene expression and response [6]. On the other hand, aberrant NRF2 activation in cancer cells confers resistance to anticancer drugs and radiation [8].
Furthermore, some additional viewpoints about Nrf2 can be inferred from the context:
Role in antioxidant defense: NRF2 plays a crucial role in antioxidant defense by regulating the expression of SLC7A11, which enhances antioxidant defense through glutathione synthesis [7]. NRF2 also contributes to the antioxidant function and detoxification in normal quiescent cells [9].
Role in metabolism: In addition to its antioxidant function, NRF2 contributes to metabolic reprogramming in proliferating cells, facilitating the pentose phosphate pathway and serine synthesis [9].
Autophagy and Nrf2 activation: Impaired autophagy can lead to accumulation of p62, which sequesters Keap1, resulting in the release of Nrf2 and increased antioxidant capacity [10].
These viewpoints showcase the various roles and implications of Nrf2 activation in cellular processes such as antioxidant defense, metabolism, and cancer cell resistance.
Figure [1]

Figure [2]
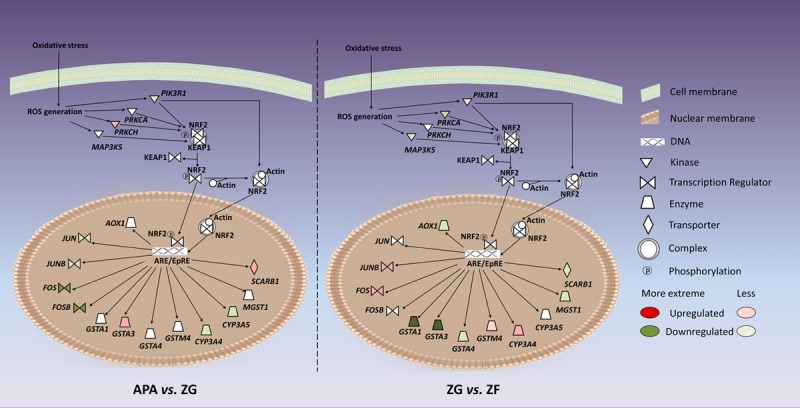
Figure [3]
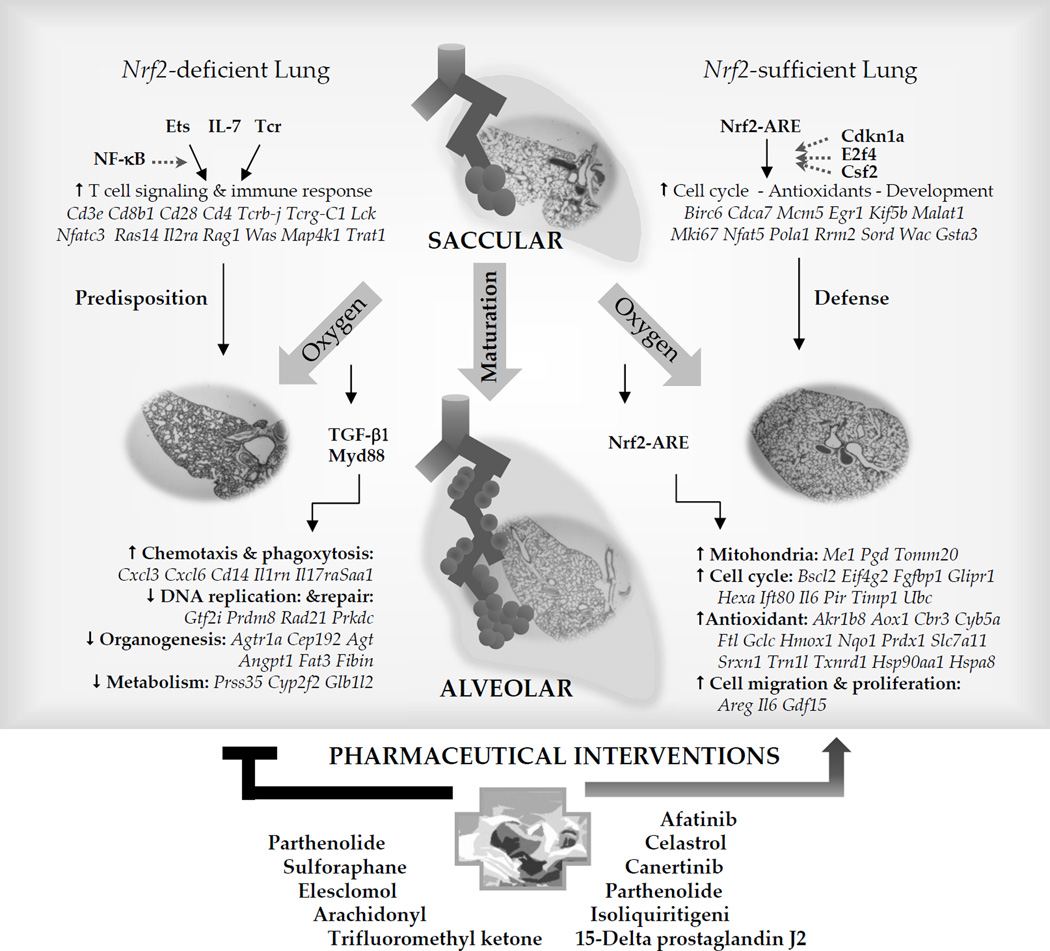
Figure [4]
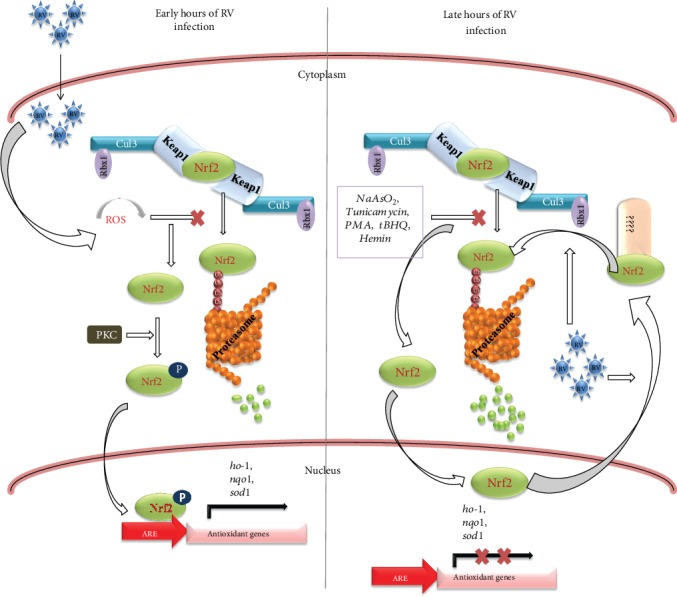
Figure [5]

Figure [6]

Figure [7]

Figure [8]

Figure [9]

Figure [10]

Note: If you are interested in the full version of this target analysis report, or if you'd like to learn how our AI-powered BDE-Chem can design therapeutic molecules to interact with the NFE2L2 target at a cost 90% lower than traditional approaches, please feel free to contact us at BD@silexon.ai.
More Common Targets
ABCB1 | ABCG2 | ACE2 | AHR | AKT1 | ALK | AR | ATM | BAX | BCL2 | BCL2L1 | BECN1 | BRAF | BRCA1 | CAMP | CASP3 | CASP9 | CCL5 | CCND1 | CD274 | CD4 | CD8A | CDH1 | CDKN1A | CDKN2A | CREB1 | CXCL8 | CXCR4 | DNMT1 | EGF | EGFR | EP300 | ERBB2 | EREG | ESR1 | EZH2 | FN1 | FOXO3 | HDAC9 | HGF | HMGB1 | HSP90AA1 | HSPA4 | HSPA5 | IDO1 | IFNA1 | IGF1 | IGF1R | IL17A | IL6 | INS | JUN | KRAS | MAPK1 | MAPK14 | MAPK3 | MAPK8 | MAPT | MCL1 | MDM2 | MET | MMP9 | MTOR | MYC | NFE2L2 | NLRP3 | NOTCH1 | PARP1 | PCNA | PDCD1 | PLK1 | PRKAA1 | PRKAA2 | PTEN | PTGS2 | PTK2 | RELA | SIRT1 | SLTM | SMAD4 | SOD1 | SQSTM1 | SRC | STAT1 | STAT3 | STAT5A | TAK1 | TERT | TLR4 | TNF | TP53 | TXN | VEGFA | YAP1

