MAPK8 Target Analysis Report Summary


About the Target
Based on the provided context information, the JNK pathway, which is synonymous with MAPK8, is involved in various biological processes and diseases. In the context of viral infections, such as DENV2, ZIKV, and CHIKV infections of salivary glands, the JNK pathway is shown to be regulated by these viruses [1]. Furthermore, in cancer cells, the Warburg effect, characterized by increased glucose uptake and glycolysis, desensitizes cells to oxidative stress-induced apoptosis by inhibiting JNK activation [2].
In the reprogramming of normal adipose tissue macrophages and those in obesity, the JNK pathway is activated to induce proinflammatory gene expression and reprogram macrophages to the M1 phenotype [3]. It is also suggested that the JNK pathway interacts with beta1 integrin signaling, affecting cell cycle regulation and radiation-induced cell cycle modifications [4].
In the context of myeloproliferative neoplasms (MPN), the JNK pathway is activated in MPN cells in response to chemotherapeutic agents, allowing the cells to escape apoptosis and prolong disease progression [5].
Overall, the JNK pathway plays a role in viral infections, cancer cell survival, macrophage reprogramming, and MPN cell resistance to chemotherapy.
Based on the provided context information:
JNK1 and JNK2 play differential roles in various skin cancers. In basal cell carcinoma (BCC), JNK1/2 enhances the interaction of Jun/Fos and phosphorylated ATF2, promoting SHH/Gli-induced tumorigenesis. In squamous cell carcinoma (SCC), JNK1 induces apoptosis while JNK2 promotes carcinogenesis in an AP1-dependent manner. SCCA1 inhibits JNK1, promoting SCC, while CYLD inhibits JNK2/AP1 cascade, suppressing SCC [6].
JNK activation is involved in apoptotic pathways. Upon apoptotic stimuli, JNK is phosphorylated and activated. JNK phosphorylates wild-type p53, leading to the formation of a complex with p73. This complex then drives the transcription of PUMA and Bax genes, ultimately causing apoptosis. However, in the case of mutant p53, phosphorylation by JNK enhances complex formation with p73 but fails to transcribe PUMA and Bax genes, reducing the apoptotic signal and promoting tumor survival [7].
ws-Lynx1 activates JNK as part of the signaling cascade. The interaction of ws-Lynx1 with alpha7-nAChRs triggers the activation of JAK, JNK, p38alpha, Src, PI3K, and AKT kinases. This subsequently activates transcription factors, including the proapoptotic factor p53, and causes mislocalization of the p27 protein. The overall effects of ws-Lynx1 are mediated by various membrane receptors and regulators [8].
JNK and p38 are activated by the IL-1 signaling pathway. Upon IL-1 binding to its receptor, the signaling module involving MyD88, IRAK, and TRAF6 is formed. This activates the MAP kinase signaling module, leading to the activation of JNK and p38 via Map kinase kinases (MKKs). The phosphorylation of JNK and p38, along with the expression of their target transcription factors cJun and cFos, results in increased IL-6 gene transcription in immature enterocytes. Later in enterocyte development, JunD replaces cJun/cFos dimers, resulting in lower IL-6 gene transcription. Additionally, a JNK-independent pathway involving JunB represses IL-6 gene transcription in the colon [9].
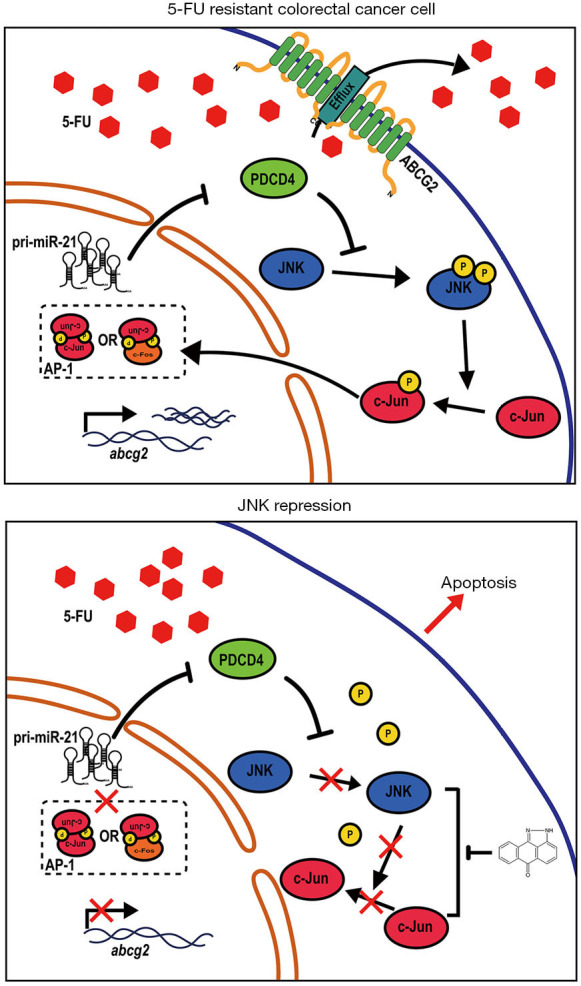
The miR-21/PDCD4/JNK/ABCG2 axis is involved in 5-FU-resistant colorectal cancer (CRC) cells. This axis represents a molecular mechanism where miR-21 regulates PDCD4, which then affects JNK signaling and contributes to ABCG2-mediated drug resistance in CRC cells. Treatment with SP600125, a JNK inhibitor, can potentially reverse the resistance in HCT116/FUR cells [10].
In summary, JNK1 and JNK2 have distinct roles in skin cancers, where JNK1 enhances tumorigenesis in BCC and induces apoptosis in SCC, while JNK2 promotes carcinogenesis in SCC [6]. JNK activation is involved in apoptotic pathways, with its phosphorylation playing different roles in wild-type and mutant p53 [7]. ws-Lynx1 activates JNK along with other kinases, leading to various cellular effects [8]. The IL-1 signaling pathway activates JNK and p38, influencing IL-6 gene transcription in immature enterocytes and repressing it in the colon [9]. The miR-21/PDCD4/JNK/ABCG2 axis contributes to drug resistance in CRC cells [10].
Figure [1]

Figure [2]

Figure [3]
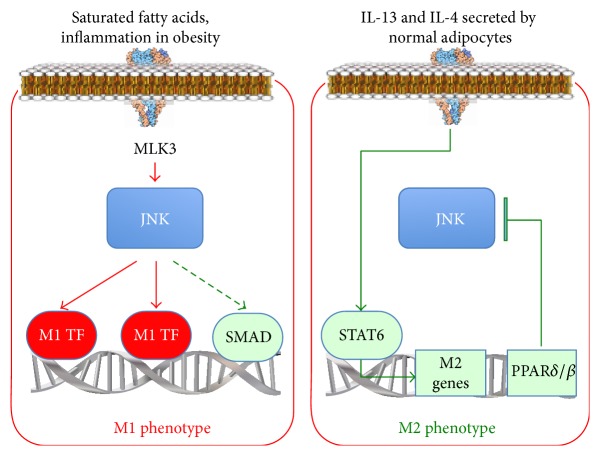
Figure [4]
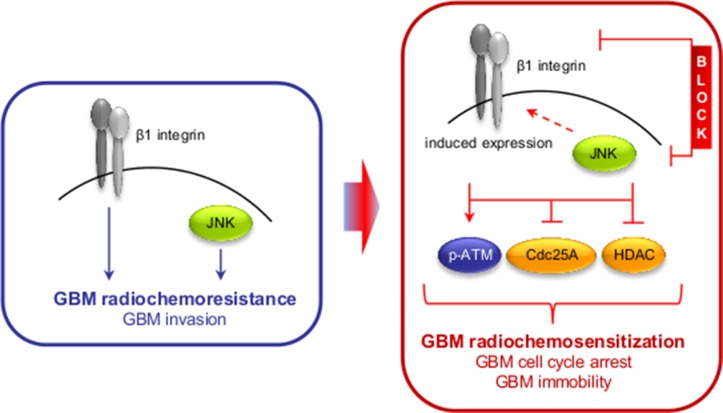
Figure [5]

Figure [6]
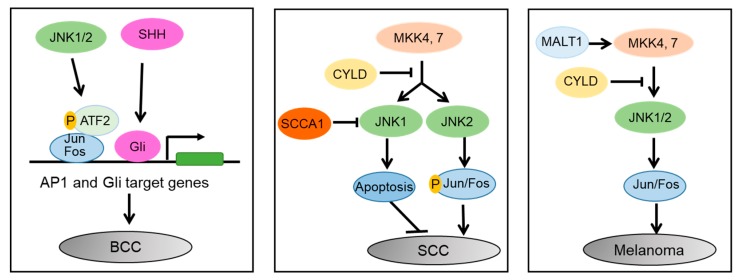
Figure [7]
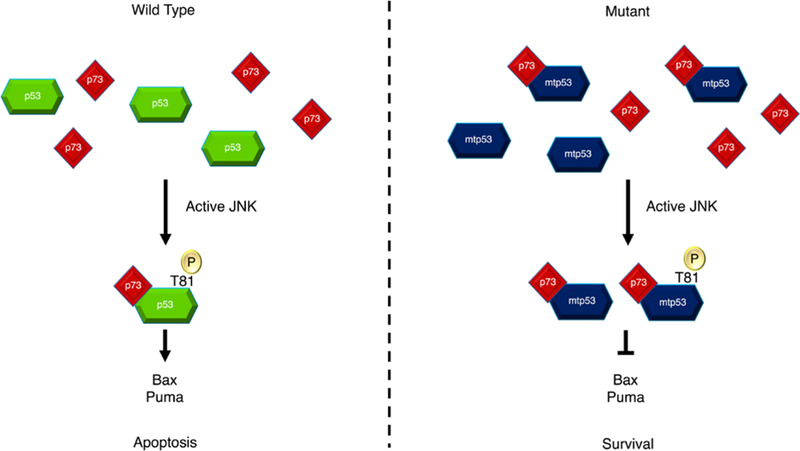
Figure [8]

Figure [9]

Figure [10]
Note: If you are interested in the full version of this target analysis report, or if you'd like to learn how our AI-powered BDE-Chem can design therapeutic molecules to interact with the MAPK8 target at a cost 90% lower than traditional approaches, please feel free to contact us at BD@silexon.ai.
More Common Targets
ABCB1 | ABCG2 | ACE2 | AHR | AKT1 | ALK | AR | ATM | BAX | BCL2 | BCL2L1 | BECN1 | BRAF | BRCA1 | CAMP | CASP3 | CASP9 | CCL5 | CCND1 | CD274 | CD4 | CD8A | CDH1 | CDKN1A | CDKN2A | CREB1 | CXCL8 | CXCR4 | DNMT1 | EGF | EGFR | EP300 | ERBB2 | EREG | ESR1 | EZH2 | FN1 | FOXO3 | HDAC9 | HGF | HMGB1 | HSP90AA1 | HSPA4 | HSPA5 | IDO1 | IFNA1 | IGF1 | IGF1R | IL17A | IL6 | INS | JUN | KRAS | MAPK1 | MAPK14 | MAPK3 | MAPK8 | MAPT | MCL1 | MDM2 | MET | MMP9 | MTOR | MYC | NFE2L2 | NLRP3 | NOTCH1 | PARP1 | PCNA | PDCD1 | PLK1 | PRKAA1 | PRKAA2 | PTEN | PTGS2 | PTK2 | RELA | SIRT1 | SLTM | SMAD4 | SOD1 | SQSTM1 | SRC | STAT1 | STAT3 | STAT5A | TAK1 | TERT | TLR4 | TNF | TP53 | TXN | VEGFA | YAP1

